World Bleeding Disorders Registry
The WBDR is the only global registry collecting standardized clinical data on people with hemophilia (PWH) and people with von Willebrand disease (VWD). The WBDR provides a web-based data entry platform to a large network of participating hemophilia treatment centres (HTCs) to collect and manage their data.
For the past five years, the WBDR has been collecting data on PWH. In early 2023, the WFH launched the WBDR VWD module, making it the first global registry to collect data on VWD diagnosis and management, and on the health outcomes of people living with VWD. WBDR data are invaluable for evidence-based advocacy, research, and improving clinical practice.
IMPACT REPORT
WBDR Impact Report 2025
The WBDR Impact Report 2025 highlights the impact, growth, and real-world value of the WBDR. It is designed to showcase the outcomes of the data, programs, and trainings over the past year. This report highlights the importance of collecting, interpreting and disseminating data in the WFH effort to improve the standard of care for people with bleeding disorders.
PARTICIPATE IN THE WBDR
The WBDR is a key data collection tool improving the provision of care for people with bleeding disorders around the world.
Pathways to improving patient care with WBDR data

HTCs interested in participating in the WBDR need to meet the following criteria:
- Have access to the internet to connect to the web-based data entry system
- Have an identified person who can input confidential patient data in the registry
- Be willing to enrol patients in the registry over the long-term and complete follow up questionnaires at each patient visit
If your HTC is interested in participating in the WBDR, please email us at [email protected]. The WFH will guide you through the process of obtaining ethics approval from your local institution or ministry of health.
Once HTCs have obtained ethics approval, the individual(s) who will be doing data entry must take a WBDR data entry training before they can start enrolling patients.
The International Data Integration Program helps to combine resources from existing bleeding disorders registries and maximize the utility of data that currently exist by facilitating data transfer from existing patient registries to the WBDR. The program is available to interested countries who wish to join this global initiative by sharing their national data and having their data represented in the WBDR. Contact the WFH at [email protected] for more information.
Current program collaborations
- Czech National Haemophilia Programme Registry (CNHPR): The CNHPR collects national data from eight pediatric and eight adult hemophilia treatment centres in the Czech Republic. As part of a proof-of-concept study, a de-identified minimal set of data from the 2018 CNHPR data were imported into the WBDR. Starting in 2019, and following the success of the first import, data on genetic testing, the Hemophilia Joint Health Score (HJHS), comorbidities, and hospitalizations were added to the imported dataset. Data from CNHPR continue to be annually updated in the WBDR.
- Hereditary Bleeding Disorders Registry (HBDR) of the Thai Society of Hematology: In 2021, the HBDR of the Thai Society of Hematology (TSH) collaborated with the WBDR to integrate their 2020 and 2021 data in the global registry. As a result of the cooperative work between the HBDR of the TSH and the WBDR, the minimum and extended data sets of 300 people with hemophilia A and B have been transferred to the WBDR as part of the latest import. More detailed information of data transfer from the HBDR to the WBDR can be obtained here (in English only).
Participating in the WBDR allows people with bleeding disorders (PWBDs) to play an active role in building this important resource and improving the future of global bleeding disorders care. If you are a PWBD, or a caregiver, and are interested in enrolling in the WBDR, please contact your HTC and inquire about their participation in the WBDR.
WBDR DATA
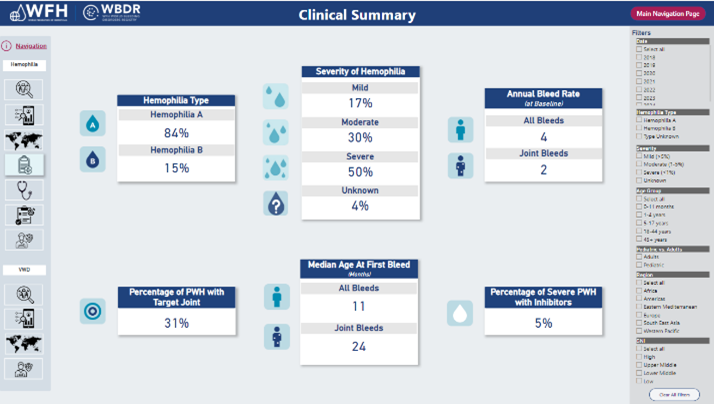
The WBDR data dashboard provides a comprehensive, visual overview of important data and metrics to enhance the understanding of the level of care and treatment in people with hemophilia and von Willebrand’s Disease around the world.
* Please note, the dashboard is currently only optimized for desktop screens. We recommend you use the dashboard on a laptop or desktop computer
The WBDR was launched in 2018 with the hemophilia data set. In early 2023, the VWD data set was added and the WBDR started enrolling people with VWD. The registry includes five functional scales and a quality of life scale.
All patient information stored in the WBDR is de-identified and confidential. The data policy guidelines of Health Solutions adhere to both the CE-mark (Conformité Européenne) and the U.K. IGSoC (Information Governance Statement of Compliance) standards, and are compliant with the General Data Protection Regulation (GDPR).
The WBDR 2023 Data Report includes data on 14,342 people with hemophilia and VWD from 119 hemophilia treatment centres (HTCs) and 44 countries. Please credit the WFH when WBDR data are used in presentations, publications, or other research materials.
Previous reports:
HTC SUPPORT
The WFH offers three WBDR programs to help HTCs collect quality data and conduct research studies using WBDR data.
WBDR Data Quality Accreditation Program
The WBDR offers a comprehensive Data Quality Accreditation (DQA) Program. The Program includes a robust data cleaning and validation process that is used to enhance the data completeness, accuracy, and consistency. The WBDR data quality team works with all HTCs, providing training and feedback on the quality of collected data.

WBDR Research Support Program (RSP)
The WBDR Research Support Program (RSP) is designed to motivate and support WBDR HTCs to collect, analyze, and use their WBDR data. The goal is to encourage countries with limited data collection experience to participate in the WBDR and use their data for research and advocacy.
WBDR HTC Funding Program (HFP)
The WBDR HTC Funding Program (HFP) is designed to provide funds to support data collection activities at WBDR HTCs in low and lower-middle income countries. The Program aims to help HTCs improve patient enrolment and improve the quality and completeness of entered data.
WBDR Manuals and Login
For more information on data entry in the WBDR, participating HTCs are invited to consult the documents below.
myWBDR APP

myWBDR is a mobile application designed for individuals diagnosed with hemophilia and/or von Willebrand disease (VWD) who are enrolled in the World Bleeding Disorders Registry (WBDR). It empowers patients by offering them a user-friendly platform to effortlessly track and monitor bleeding episodes, pain levels, treatments, and overall health status using EQ-5D-5L and PROBE questionnaires.
FAQ
If you have any questions, we invite you to check the WBDR FAQs or contact us at: [email protected]
CONTACT US
Please contact the WBDR team at [email protected].

WBDR HTC Funding Program
Provides funding for data entry activities at participating HTCs

WBDR 2023 Data Report
THANK YOU
To each person with a bleeding disorder who has kindly agreed to share their data: thank you for helping improve the quality of care for people with bleeding disorders (PWBDs) around the world.
To each of the WBDR HTCs: thank you to all of the dedicated staff at participating HTCs who work hard to ensure that their data is meeting WBDR data quality standards.
The WFH thanks all of our sponsors for their generous financial support which allows us to continue to develop this important initiative. The WBDR is supported by funding from:
VISIONARY PARTNER

COLLABORATING PARTNERS



Did you know?
The WFH PACT Program is a 5-year initiative designed to improve outreach and diagnosis and increase access to sustainable care for people with inherited bleeding disorders.
Stay informed
Keep up to date on the latest developments in the bleeding disorders community, from medical advances to important events.